Minimally Invasive Morcellation Device
Overview
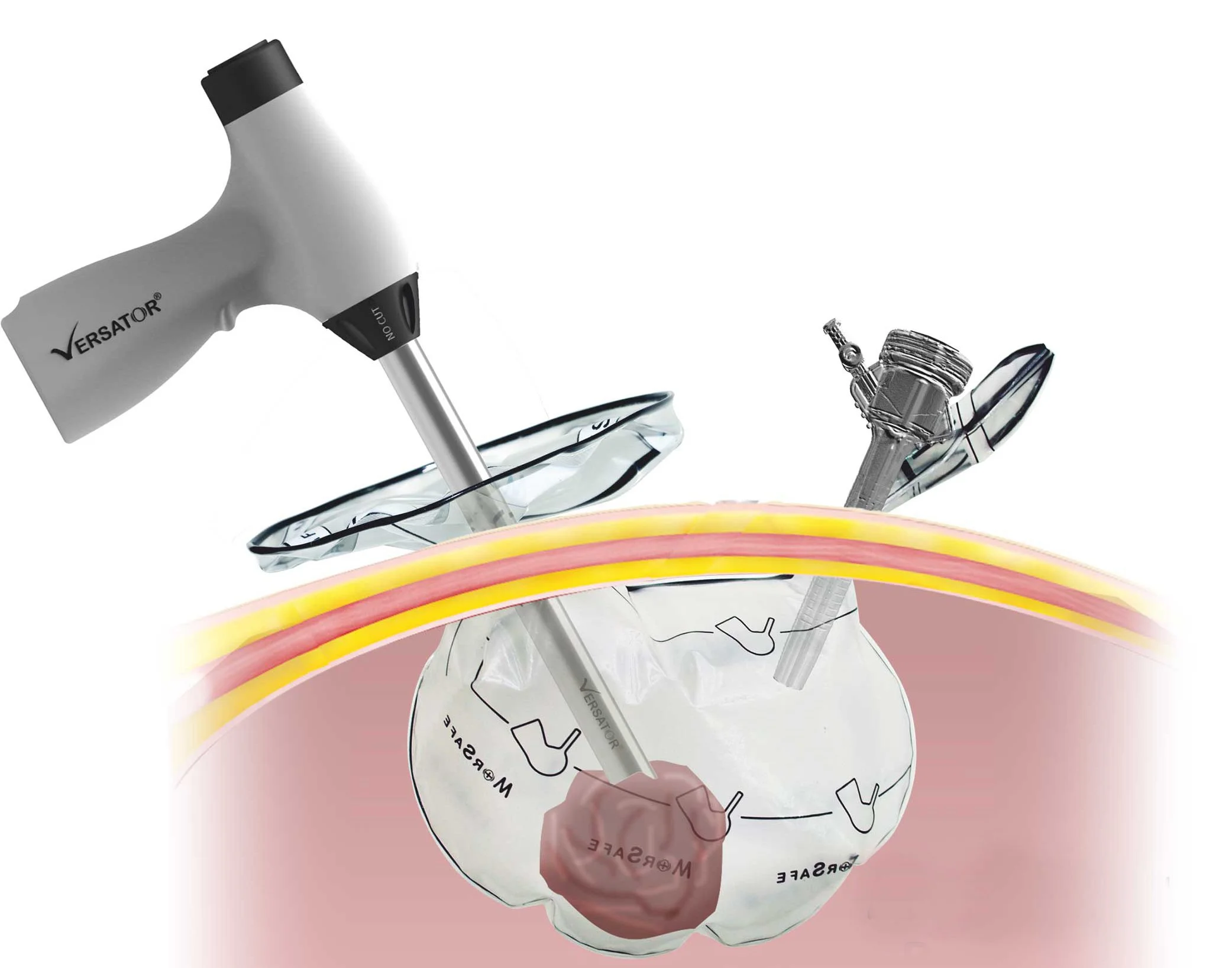
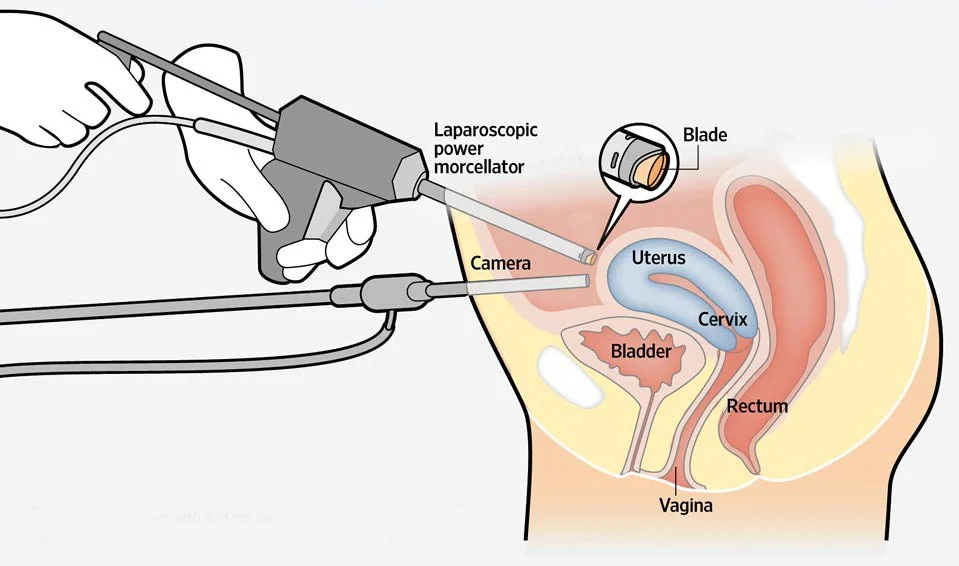
Design and development of a compact surgical morcellation device, focused on miniaturisation, improved sterilisation, and compliance with medical device standards for safe and effective use in minimally invasive procedures.
Client / Sector
Medical – Surgical Instrumentation
Versator
The Challenge
Redesign the device envelope to achieve further miniaturisation of the mechanical system while improving ease of sterilisation. The solution needed to align with relevant medical device standards, including biocompatibility, safety, and reprocessing requirements, while supporting third-party validation and certification.
Our Approach
Led the product development with a focus on compact mechanical integration and design for sterilisation, ensuring alignment with standards such as ISO 13485 (quality management), ISO 10993 (biocompatibility), and IEC 60601 considerations where applicable. The design reduced internal complexity to support effective cleaning and reprocessing.
Coordinated closely with suppliers and the client—fully remotely—to prepare the product for independent third-party testing, including validation of sterilisation processes, material compliance, and mechanical performance.
Solution Delivered
A refined, miniaturised morcellation device featuring an optimised mechanical architecture and a sterilisation-friendly design, developed in line with medical regulatory expectations and ready for third-party verification and certification.
Outcome / Impact
Delivered a compact and clinically practical solution that improved handling and sterilisation workflows while aligning with medical compliance pathways. Successful engagement with third-party testing processes ensured the product met required safety, performance, and regulatory standards.
Scope of Involvement
End-to-end development including concept design, mechanical integration, compliance consideration, supplier coordination, and support through validation and third-party testing.
Key Capabilities Demonstrated
Medical device design (ISO 13485 alignment)
Design for sterilisation & reprocessing
Biocompatibility-aware engineering (ISO 10993)
Third-party testing and validation coordination
Precision mechanical integration
Remote project leadership